Making of PSA
Contents
- 1 Making of PSAs
- 1.1 Effect of important parameters on PSA making and performance
- 1.1.1 Effect of polymer molecular weight and crosslinking reactions on the end-use properties of PSAs
- 1.1.2 Effect of composition on Mechanical behaviours and fracture energy of PSAs
- 1.1.3 Effect of tackifier on PSAs
- 1.1.4 Effect of chain transfer agent and cross-linker concentration in making of PSAs
- 1.1.5 Effect of flexible substrates on PSAs performance
- 1.1 Effect of important parameters on PSA making and performance
- 2 PSA performance measurement
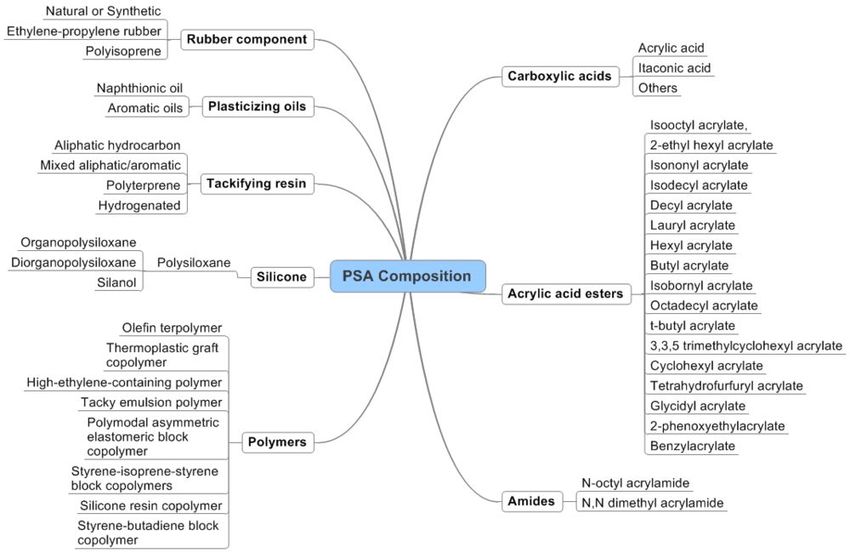
- 3 PSA Composition
Making of PSAs
Although PSAs can be obtained by different polymerization processes (i.e., emulsion, solution, hot-melt, or radiation curing), much attention has recently been devoted to the utilization of more environmentally friendly processes such as emulsion polymerization. Soft polymer networks are commonly used as previous termpressure sensitive adhesivesnext term (PSAs). This is due to their unique ability to deform and yet to resist flow. These contradictory requirements indicate that the mechanical properties are finely tuned, and that the types of deformation upon application are carefully considered. Variety of PSAs can be prepared by mixing a linear vinyl terminated polymer with a silane terminated f-functional cross-linker. Jensen et al., 2009
| Sr. No. | PSA process | Chemical composition | Time of launch |
| 1 | Solvent-based | Rubber/resin, acrylics, silicones | Since 19th century |
| 2 | Hot-melt | Block copolymers, acrylics | 1940s |
| 3 | Emulsion (water)-based | Acrylics, natural and synthetic rubber, ethylene-vinyl acetate copolymer | 1970s |
| 4 | Radiation-cured | Acrylics, rubber | 1970s |
| Sr. No. | Properties | Solvent-based: acrylic | Hot-melt: styrene-isobutylene-styrene. | Emulsion based: acrylics |
| 1 | PS performance | Excellent | Excellent | Very good |
| 2 | Ease of compounding | Moderate | Difficult | Easy |
| 3 | Formulation flexibility | Limited | Excellent | Moderate |
| 4 | Coating method flexibility | Limited | Poor | Excellent |
| 5 | Ease of changeover | Limited | Poor | Excellent |
| 6 | PSA reproducibility | Excellent | Limited | Excellent |
| 7 | Aging properties | Excellent | Poor | Excellent |
| 8 | Clarity/color | Excellent | Poor | Excellent |
| 9 | Safety/toxicity | Poor | Poor | Excellent |
| 10 | Raw material costs | High | Low | Medium |
| 11 | Coating/compounding costs | High | Medium | Low |
Effect of important parameters on PSA making and performance
Effect of polymer molecular weight and crosslinking reactions on the end-use properties of PSAs
In a study wherein polymer molecular weight and polymer microstructure were regulated using different chain transfer agent (CTA) concentrations and by addition of a diacrylic monomer (MM) it was shown that all of the measured adhesion properties strongly depend on molecular weight of the synthesized polymer and on the amount of gel phase Kajtna et al., 2009
Effect of composition on Mechanical behaviours and fracture energy of PSAs
In a study it was shown that the mechanical behaviour depend on their composition but majority of fracture energy is dissipated on the first millimetre near the bending zone where fibrils elongation is maximum. Observations of interfaces between PSAs and glass substrate underline that fracture energy varies linearly according to the contact area Horgnies et al., 2007
Effect of tackifier on PSAs
To study the effect of tackifier (such as hydrogenated cyclo-aliphatic resin) a model system consisting of polystyrene-b-polyisoprene-b-polystyrene triblock copolymer was prepared. Tackifier increased the peel adhesion significantly and the increase became stronger above 40 wt% tackifier. The higher peel adhesion was obtained in the system with the larger amount of agglomerates of tackifier in the polyisoprene matrix. Sasaki et al., 2008
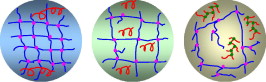
Effect of chain transfer agent and cross-linker concentration in making of PSAs
In a study it was shown that a constant cross-linker concentration, one can manipulate the polymer micro-structure by adding varying amounts of chain transfer agent. Three examples of these micro-structures are depicted below which show a tight gel network with long-chain sol polymers, a loose gel network with shorter sol polymers, and an imperfect gel structure with highly branched sol polymers. By manipulating the micro-structure, previous termpressure-sensitive adhesivenext term performance can be affected. Qie and Dube, 2010
Effect of flexible substrates on PSAs performance
The fracture energy (fracture toughness) of tapes during globally elastic unpeeling is often calculated from the relation G=P/b(1−cos θ). A study suggested that this expression is correct for elastic peeling from rigid substrates but it gives misleading results when peeling from reversible flexible substrates. Steven-Fountain et al., 2002
PSA performance measurement
PSAs polymeric materials effect tack, peel and shear strength . Inherent properties such as copolymer composition and microstructure, molecular weight and distribution are among the most influential factors affecting PSA properties directly as well as indirectly through their influence on physical properties (e.g., the glass transition temperature, Tg) and thus, rheological properties of the polymer (e.g., viscoelastic regions, moduli).
Therefore, PSA is the result of a fine balance between these three major, interrelated properties.
Tack
It is a measure of the force required to remove, say a foam gasket and its adhesive, from the substrate. It usually refers to the initial attraction of the adhesive to the substrate. Tack can be measured by four basic methods these are loop tack, rolling ball, Quick stick and probe measurement devices. Review Of Methods For The Measurement Of Tack
Peel strength
Peel strength is measured as a force required to remove a standard PSA strip from a specified test surface under a standard test angle (e.g., 90° or 180°) under standard conditions. Much like tack, manufacturers control adhesion to create different products based on user requirements. After a PSA has been applied to the substrate, adhesion continues to increase for a period of time — typically 24 hr.
Shear strength
Shear strength is the internal or cohesive strength of the adhesive mass. Usually, it is determined as the length of time it takes for a standard strip of PSA to fall from a test panel after application of a load. Usually, tack and adhesion decrease as shear strength increases. Emulsion-Based Pressure-Sensitive Adhesives: A Review